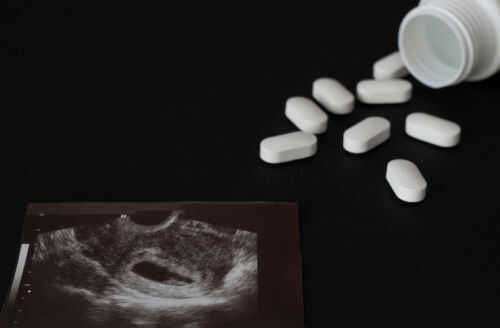
One Supreme Court justice just hit pause on a lower-court move that would have stopped abortion pills from being shipped to women’s mailboxes nationwide.
Quick Take
- Justice Samuel Alito issued an administrative stay that temporarily restores the FDA’s 2023 rule allowing mifepristone to be prescribed via telehealth and sent by mail.
- The stay blocks a recent Fifth Circuit order that had temporarily halted remote prescribing and mail delivery, creating immediate uncertainty for providers and patients.
- Louisiana’s lawsuit is moving forward after the Fifth Circuit said the state has standing tied to Medicaid costs for emergency-room care following reported complications.
- The stay is time-limited and is set to expire May 11, 2026, while the Supreme Court considers next steps and receives responses from Louisiana.
What Alito’s temporary order actually changes—and what it doesn’t
Justice Samuel Alito’s administrative stay restores, for now, the FDA’s 2023 framework that allows mifepristone to be prescribed through telehealth and dispensed through the mail or at pharmacies without an in-person doctor visit. The stay does not decide the underlying case on the merits. It simply prevents the Fifth Circuit’s temporary restriction from taking effect while the Supreme Court evaluates emergency requests from the drugmakers.
The practical effect is a brief window of continuity for abortion providers and patients who rely on remote prescribing. That continuity matters because healthcare systems tend to change protocols quickly when legal rules swing. Even when court orders last only days, clinics must decide what forms to use, what disclosures to provide, and how to avoid dispensing a medication under a rule that could change again with little notice.
The legal fight turns on standing, federal power, and state costs
The Fifth Circuit’s recent order carried weight not only because it restricted mail delivery, but because the panel unanimously said Louisiana had legal standing to sue. The court pointed to the state’s claim that its Medicaid program covered emergency-room care for two women who experienced complications after receiving mifepristone from an out-of-state provider. That standing finding distinguishes this lawsuit from a 2024 challenge the Supreme Court rejected unanimously for lack of standing.
Under the surface is a larger federalism question conservatives have argued about for years: how far an executive-branch agency can go in setting nationwide rules that shape healthcare delivery, and when states can contest those rules based on real-world costs. The FDA’s rulemaking on mifepristone expanded access step-by-step starting in 2016, accelerated during the COVID-19 era, and was formalized in 2023 after the agency concluded the drug may be safely used without in-person dispensing.
Why this case is a flashpoint for “government trust” on both sides
Abortion fights often get framed as red versus blue, but this dispute also taps into a broader skepticism about institutions that crosses party lines. Supporters of the FDA’s approach argue the agency evaluated safety and modernized access through telehealth and pharmacies. Critics argue that removing in-person safeguards shifts risks onto women and pushes downstream costs onto states and taxpayers, especially when complications lead to emergency care reimbursed by Medicaid.
Louisiana officials have made that critique in sharp terms, including a statement from the state attorney general attacking the drugmakers’ motives and warning of reduced medical oversight. Meanwhile, manufacturers Danco Laboratories and GenBioPro asked the Supreme Court for emergency relief and want the justices to review the Fifth Circuit’s order. The competing claims highlight how hard it is for citizens—left, right, and independent—to feel confident that public policy is being made transparently and with accountability.
The clock is short: what to watch before May 11
The stay runs only until 5 p.m. on May 11, 2026, meaning the legal status of mail distribution could shift again quickly. Alito also set a deadline for Louisiana officials to respond to the emergency appeals. Providers have reportedly prepared contingency plans, including asking patients to approve receiving shipments that may or may not include mifepristone, depending on how the legal process unfolds in the coming days.
It's for 1 week. Then they will decide whether to make it longer while litigation proceeds or to hear the case themselves now.
"ALITO temporarily restores FDA rule allowing abortion pill to be sent by mail"https://t.co/0dtc4OHfnj— Bruce Duck (@BruceDuck2) May 4, 2026
For conservatives who prioritize limited government and clear lines of authority, the coming week is likely to feel like another test of whether major national policy can be made through agency rules that bounce between courts. For liberals who prioritize broad access, the same timeline underscores how fragile nationwide “rights in practice” can be when they depend on administrative policy and emergency court orders. Either way, Americans are again watching big, life-altering decisions get made on a tight legal clock.
Sources:
Supreme Court Restores Access to Abortion Pill Mifepristone Through Telehealth, Mail and Pharmacies
Mifepristone: Alito Supreme Court FDA mail














